EXPERT INSIGHT
35 Years of Making a Difference: Turning Medtech Innovation into Everyday Care
When Materialise Founder Fried Vancraen started the company 35 years ago, his goal was to create a better and healthier world for all. Throughout the years, we've helped medtech businesses bring this mission to life time and time again, always striving to provide healthcare professionals with equipment that makes their lives easier and patients with a medical device designed specifically for them. Even after decades of turning trailblazing medtech innovation into everyday patient care, we're just getting started.
AM's role in life-changing medical devices
Making a difference often starts with smaller changes. For additive manufacturing (AM) in medtech, it's seen clearly through clever design. Faster time-to-market, efficient customization, lighter parts, less assembly — the advantages quickly add up. AM's impact doesn't stop at prototyping either. Certified manufacturing processes and materials that prioritize cleanability and biocompatibility ensure that innovation is maintained throughout the entire product lifecycle.
AM has brought a new level to medtech, both in day-to-day advancements and in exceptional circumstances. The latter was especially clear during the COVID-19 pandemic. The world needed medical solutions — fast. 3D printing was the natural choice, offering quick turnaround for novel applications. At Materialise, we stepped in to support urgent needs, from critical medical devices to personal protective equipment and preventative devices.
One such solution was a 3D-printed, non-invasive positive-end expiratory pressure (PEEP) mask — a rapid response to the dire shortage of mechanical ventilators. According to Prof. Wilfried De Backer, Pulmonologist and Director of respiratory imaging company FLUIDDA, who partnered with us on this development, “The aim is to avoid invasive treatments. This new solution gives positive pressure and oxygen, so it is not a big burden on the patient. You can avoid more invasive treatments and save on ventilator capacity, which you can use on other patients.”
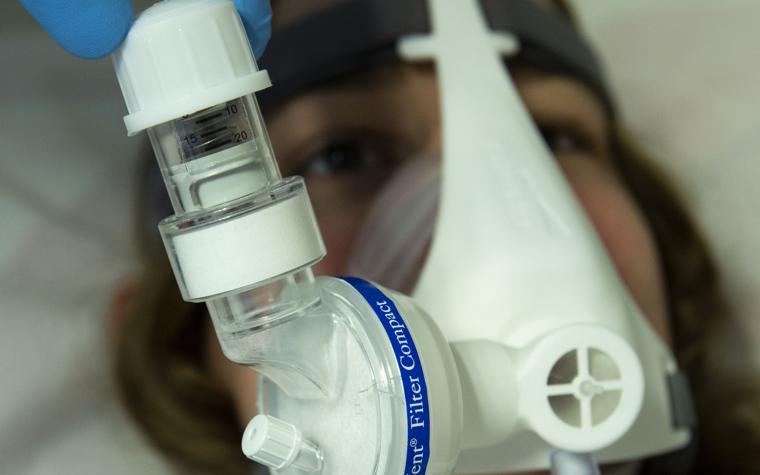

In a time when the world was in great need of life-saving medical devices and invasive ventilator treatments were placing enormous strain on patients and healthcare systems, 3D printing provided a new opportunity for hospitals and their most vulnerable patients.
AM's impact on medtech during COVID is just a small glimpse into its potential. Over the past 35 years, thousands of cases have emerged, from creating niche solutions to redefining industries.
From smart design upgrades to complete industry transformation
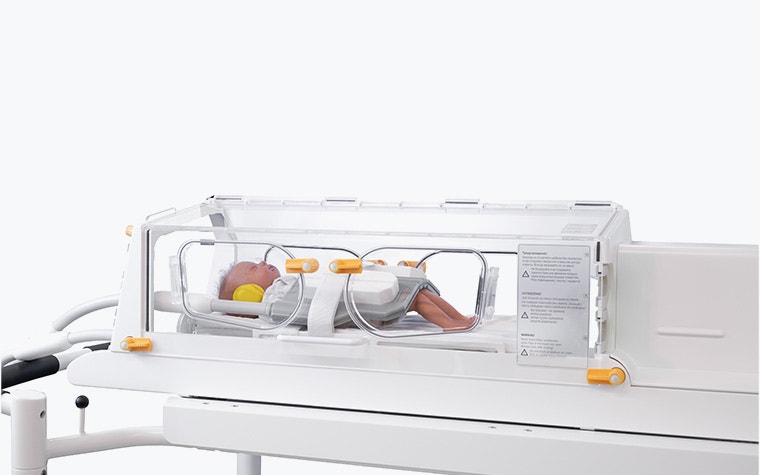
Medical devices redesigned with AM are standout examples of the technology's impact, and LMT Medical Systems offers a compelling case. Their unique incubators allow medical staff to safely transport newborns and premature babies directly from the NICU into the MR suite for an MRI. Since they only produce a small number of units each year, AM was a clear choice to avoid excessive stock. In addition to meeting all the strict requirements of MRI technology, redesigning the incubator with AM also reduced its weight by 15 kg, making a significant difference for the medical staff transporting both the device and these fragile patients.

On a broader scale, AM offered a significant contribution to the medtech industry in the year 2000. The technology's ability to easily and affordably make personalized devices at scale brought the ideal fit to nearly all patients in the hearing aid industry. What once demanded a labor-intensive production process from a limited number of skilled artisans can now be digitally personalized with AM software at scale, producing hearing aids that are more comfortable and acoustically optimized. This approach rapidly became the industry standard, and today, 99% of the world's hearing aids are manufactured with 3D printing.
Since then, other medtech segments experienced similar evolutions. Take orthotics, for example. Every patient walks with a unique gait; their orthotics should be just as unique. At Materialise Motion, we use dynamic gait analyses to create fully personalized insoles tailored to each customer's specific needs and required corrections. Demand for this level of personalization surged, and production of the insoles skyrocketed 400% in just four years.
Today, 18 of the top 20 medtech manufacturers partner with us to bring their innovations to life, and our facilities produce over 150,000 medtech parts annually. With ISO 9001-certified processes, meticulous traceability, and extensive experience with regulatory bodies worldwide, we help them navigate complex compliance requirements and bring safe, reliable medical devices to market with confidence. For Sartorius alone, we've printed more than 26,000 biocompatible, single-use components in small-series batches.
“In a highly regulated organization like ours, we have to have a good relationship with our suppliers. We have to do the vendor QA audits and ensure they’re compatible with our QMS, so you don’t want a big range of suppliers,” says Jeremy Pullin, Head of Additive Manufacturing at Sartorius. “The breadth of processes that Materialise offers, along with the credibility and experience within the industry that only comes over time, is a big draw to us.”


Engineering on anatomy
Aside from manufacturing devices in our own certified production lines, we're also committed to empowering medical device companies with advanced digital solutions. One example is the adoption of digital twins, or digital representations of real patients, via software tools like Materialise Mimics. Companies can test fast, fail fast, and adjust fast to accelerate the verification and validation stage of development. The benefits quickly become clear in practice. In some cases, early implant failure could lead to a loss of built-up inventory. However, by revealing this problem early in the design verification process with digital twins, companies can entirely avoid this loss.
“Digital twins and in silico medicine are the most promising solutions to refine, reduce, and (partially) replace animal testing and clinical trials through in silico trials," said Thierry Marchal, Industry Director for Healthcare Solutions at Ansys and Secretary General of the Avicenna Alliance. "Eventually, digital twins have the potential to predict and prevent diseases, making our lives longer and healthier.”
Beyond digital twins, our software platform supports medtech innovation at every stage, from device design to surgical planning and extended reality. Look at the structural heart field, for example, where AI-driven 3D insights are helping companies bring new therapies to market faster and more effectively.


Once a device clears verification, case numbers can boom with automated steps and a more accurate patient selection process made possible by 3D measurements. Plus, physicians are better equipped to adopt devices thanks to the intuitive, detailed 3D insights they get into implant-anatomy interactions.
One particular Fortune 500 medical device company illustrates these advantages beautifully, using Mimics' AI Heart Segmentation algorithm to create 3D anatomical models with speed. Thanks to this flow, they've ramped up their case load from one to two per month to ten per week.
Just the beginning for AM in medtech
The next chapter for medtech is all about scaling personalized care further and making it accessible to more patients than ever before. Developments in augmented reality, robotics, and biocompatible materials are poised to drive rapid innovation in medtech in the years to come. Our role is to help medtech companies tap into these advancements — guiding them through the complex regulatory landscape and providing the digital solutions and expertise that turn groundbreaking ideas into safer, more effective devices for the people who need them most.
Looking back, our technology has already reshaped entire medtech segments. Today, it’s enabling more personalized solutions to enter the market at a rapid pace. And tomorrow, it will scale to even greater heights. This is the mission we’re proud to be a part of — helping innovation move from first idea to everyday care.
Share on:
You might also like
Never miss a story like this. Get curated content delivered straight to your inbox.