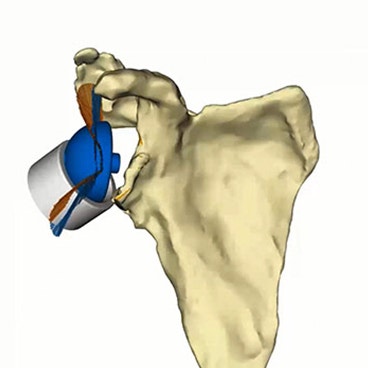
Materialise Glenius
Personalized glenoid implants
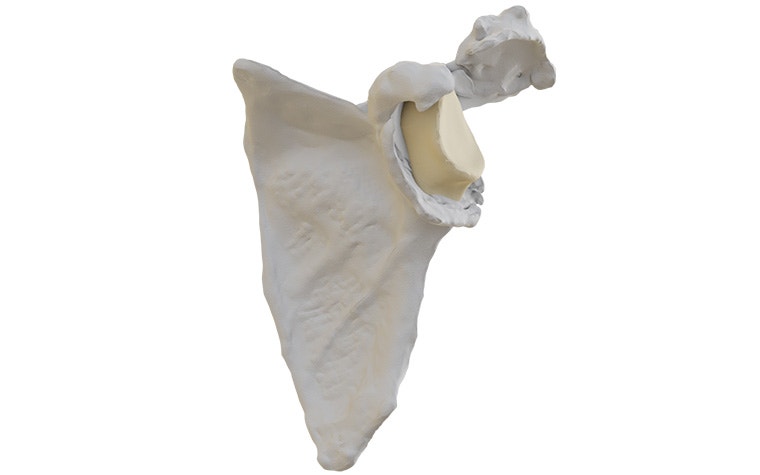
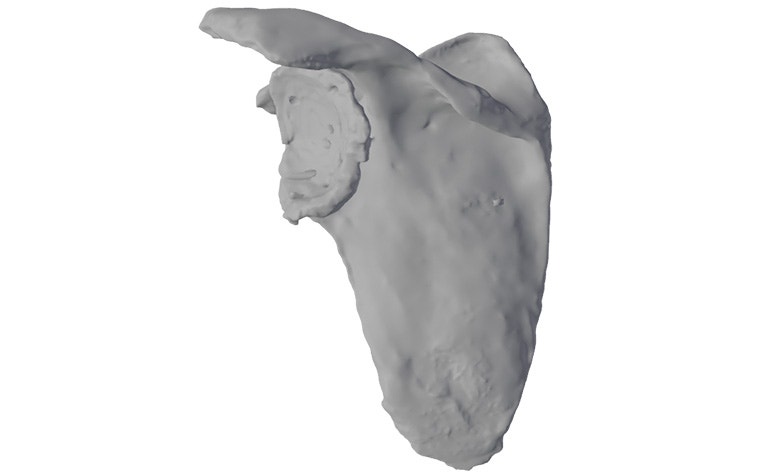
In a shoulder arthroplasty with a severe glenoid defect, the remaining bone stock is often insufficient to fixate standard baseplates, and the defect can be too large or oblique for a bone graft.
Don't compromise the outcome of complex shoulder cases. Enhance predictability with personalized Glenius implants.


Get it right first time
Trustworthy results
Rely on our precise solutions to ensure successful outcomes. Powerful 3D planning and optimized implant designs lead to a 96% survival rate.1
More than a revision implant
Most patients present Wallace type 2 and 3 defects in primary and revision cases. Leverage tailored solutions to address these complexities specifically.
Maximize bone preservation
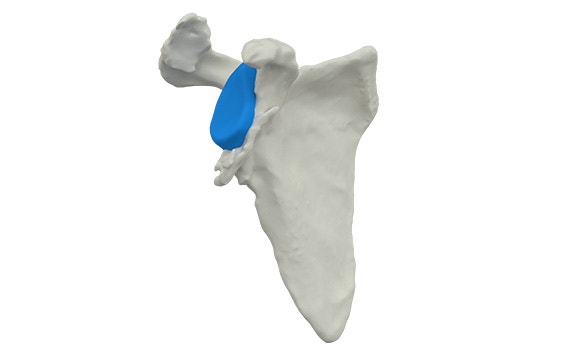
Adapt the implant to the patient instead of the other way around.
Unmatched experience
Easily collaborate with clinical engineers using an intuitive online 3D planner and benefit from our extensive expertise in 3D planning and printing.
How Glenius makes personalization easy
Unique 3D analysis
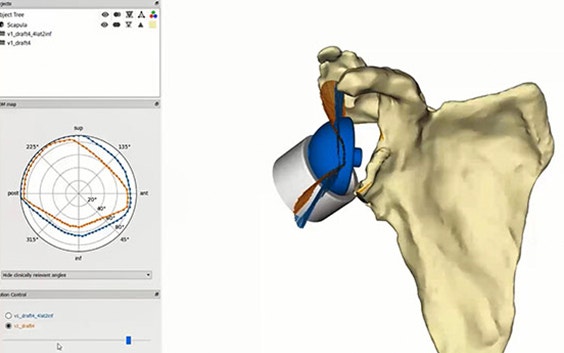
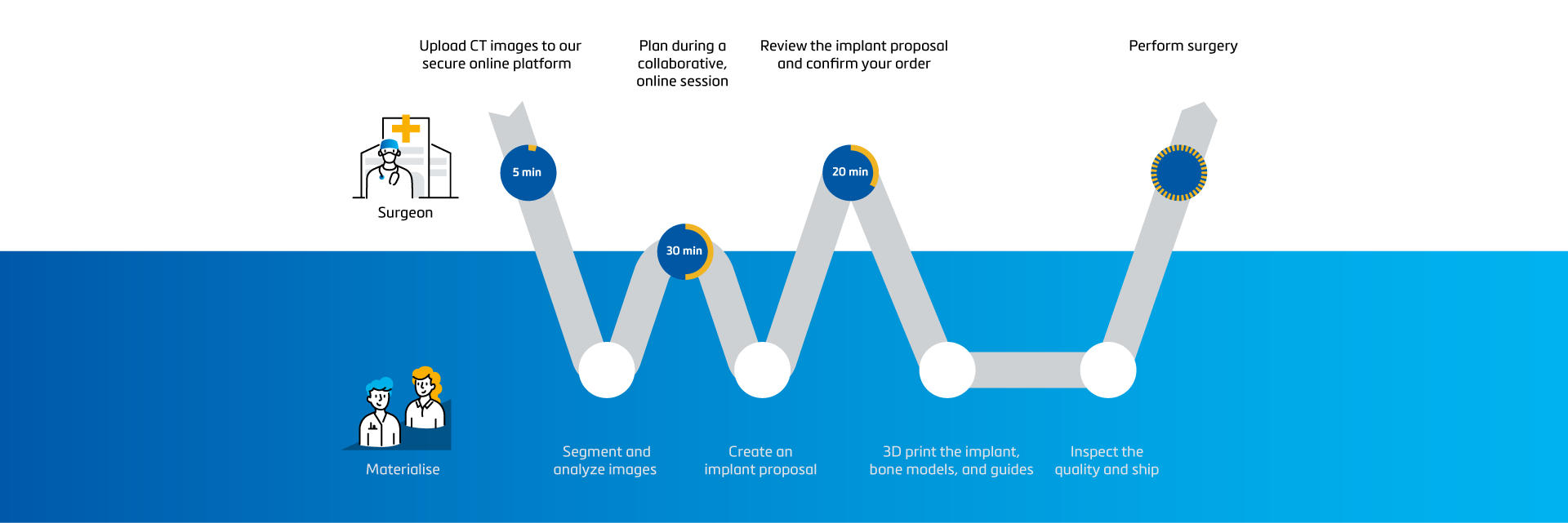
Based on the segmented CT scan, our experienced clinical engineers implement statistic shape model technology to quantify the glenoid bone loss.
Expert clinical engineers
Our clinical engineers bring 30+ years of experience with personalization to the collaboration so you don't have to invest too much time or effort into the process. They design the Glenius implants to best restore the patient's specific anatomy and maximize bone preservation as much as possible.
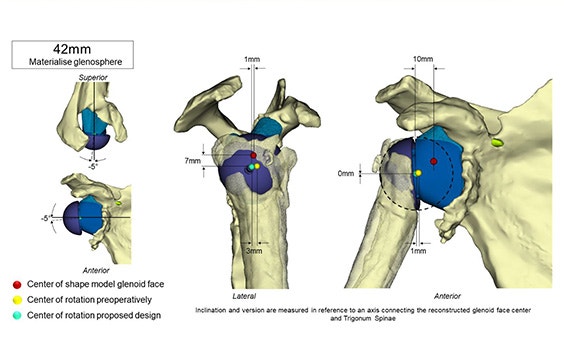
Optimized long-term fixation and stability
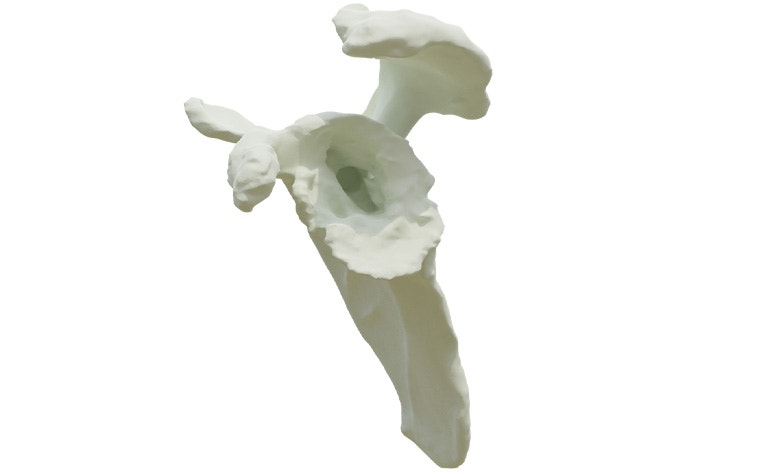
Glenius implants use crossed screw trajectories to optimize fixation. The integrated augment mimics bone properties and is designed to improve secondary fixation through bone on growth.
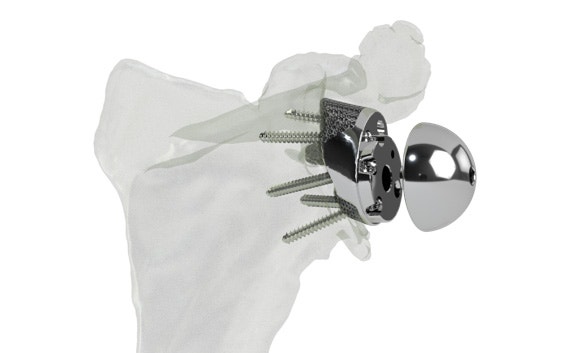
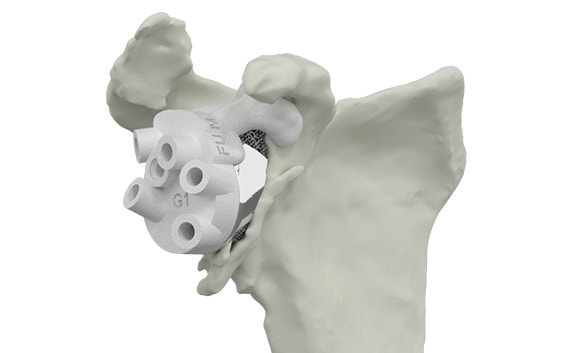
Personalized instrumentation
Innovative 3D-printed guides optimize primary fixation by directing the required cross-fixated screws as indicated by the detailed and personalized preoperative plan.
Easy-to-use digital platform
SurgiCase is an all-in-one web-based collaboration platform where you can upload images, plan, and collaborate with clinical engineers. You can securely access the platform from any internet browser.

Indications
A streamlined process with expert support

Clinical evidence
Access the latest articles supporting clinical evidence for the Glenius implant here.
Patients with severe glenoid deficiencies show adequate pain relief, reasonable functionality, and good patient satisfaction by using Glenius Glenoid Reconstructive System
Treatment of severe glenoid deficiencies in reverse shoulder arthroplasty: the Glenius Glenoid Reconstruction System experience (2019)
Promising clinical and radiological outcomes for custom-made glenoid components in revision rTSA, with no signs of loosening implants or scapular notching
Functional and Radiological Outcomes after Treatment with Custom-Made Glenoid Components in Revision Reverse Shoulder Arthroplasty (2022)
A global impact
550K+
orthopaedic patients treated
400+
Glenius implants designed
12+
years of experience
Get inspired
Discover how others benefit from the personalized Glenius implant.
Learn about Glenius and get support
Learn how to get started and see how leading experts are using the Glenius solution. If you are already a user, our customer support can help you with all your questions.
Frequently asked questions
L-102611-01
Materialise Glenius is MDR-compliant.
References:
1 Based on post-marketing surveillance (PMS) in 2019
You might also like
Orthopaedic planning software
Personalization made easy for shoulder arthroplasties
Unmatched excellence in bone deformity corrections
Go beyond visualization and interact directly with the anatomy to bring greater clarity to in-house preoperative planning.