Webinar
Advanced 3D Simulation, Modeling, and Printing in TMVR
Augment the predictability, trust the outcome
About this webinar
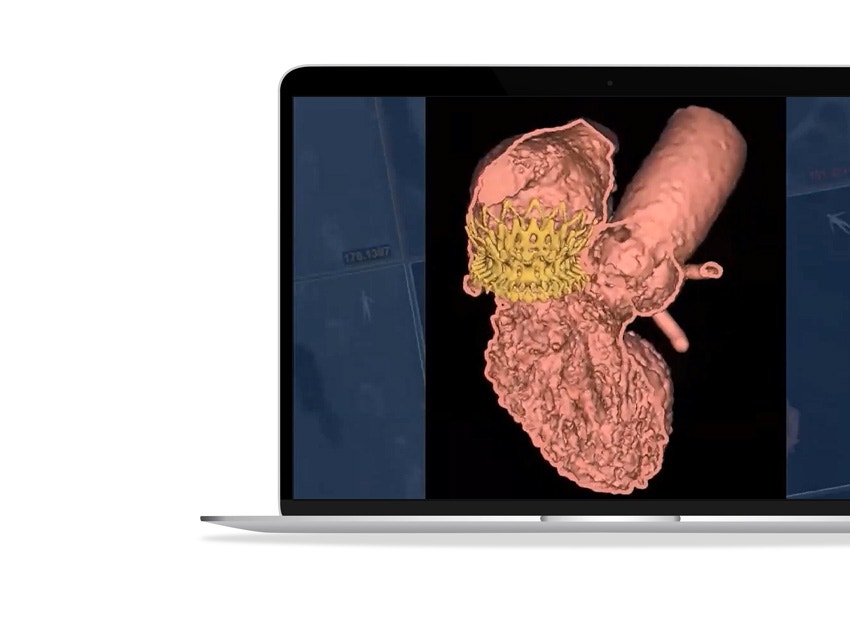
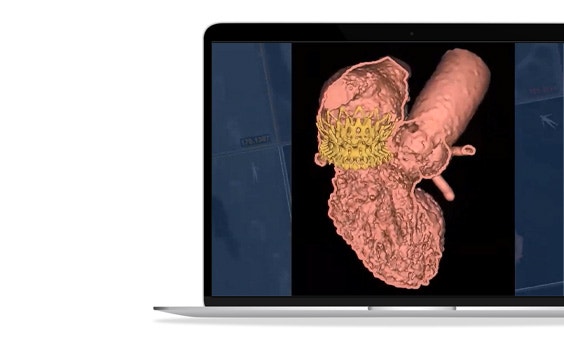
As part of the CSI virtual webinar series, Prof. Modine from the CHU de Bordeaux presented on advanced 3D simulation, modeling, and printing in TMVR.
3D-CT planning provides enhanced 3D insights, and its use of AI allows for consistent and efficient planning. Its automatic method is proven to reduce neo-LVOT assessment time by between 67% and 87%* and provide a more consistent neo-LVOT prediction (ICC= 0.99) than traditional CT planning (ICC = 0.89)**.
What you will learn
- Why TMVR is here to stay
- How to approach TMVR CT screening and device selection using Sapien, Tendyne, Intrepid, or Cephea devices
- How 3D-CT planning can impact the screen failure rate for TMVR
- The accuracy of 3D-CT planning, compared to post-op results
Speakers

Thomas Modine, MD, PhD
Share on:
*Ward et al., A More Efficient Method of Patient Evaluation for TMVR: Planning for Best- and Worst-Case Position, JACC, 2019 **Wang et al., A More Reliable Method for Predicting Neo-LVOT Obstruction after TMVR, JACC, 2018 Materialise medical device software may not be available in all markets because product availability is subject to the regulatory and/ or medical practices in individual markets. In countries where no regulatory registration is obtained of Mimics and/or 3-matic Medical, a research version is available. Please contact your Materialise representative if you have questions about the availability of Materialise medical device software in your area. L-102238-01