EXPERT INSIGHT
Personalization at Scale: How Are We Going to Increase Adoption?

By now, you have probably heard it said that personalization is the future of healthcare. Managing a company that has driven the development of personalized tech solutions for almost three decades, I would say that this future is not so far away.
Back at the beginning of our journey in the medical field, when we started with the launch of software tools to visualize medical images in 3D, we had no idea that this was going to be a milestone in personalized healthcare.
The real breakthrough was the possibilities it brought. From the moment we could visualize the anatomy in 3D, we could start looking at problems in another way and see things that would be otherwise impossible in 2D. More importantly, it also gave us the possibility to start planning based on precise information and significantly improve the predictability of patient outcomes.


Some years later, these solutions evolved into planning tools that can simulate surgeries before entering the OR. In the OR, 3D printing technology has allowed us to go the extra mile in personalization by introducing surgical guides and implants that are unique to each patient.
From the digital planning of standard procedures to the design of complex patient-specific titanium implants, personalized solutions have proven their value by bringing an extra level of understanding and guidance to surgeons. Since the launch of our Mimics software in 1993, we estimate that our planning software and the introduction of 3D-printed devices have helped more than 10 million patients, directly or indirectly.
Still, some in the industry would call it a niche. We disagree, and we have good reasons for that.
A great example is the announcement we made last year. We achieved the European CE marking of our personalized 3D-printed orthopaedic surgical guides and our entire cranio-maxillofacial (CMF) portfolio, including models, surgical guides, and metal implants.
Regulation at the level from the standard to the stand-out
This CE marking demonstrates that these personalized devices follow the same stringent requirements as standard devices, despite the challenges involved in making every device match the specific patient's anatomy. Crucially, this makes them readily available to integrate into current hospital workflows, avoiding exception procedures.


With the CE process in place, surgeons won't need to sign special prescriptions for the devices they order. This means less paperwork for the hospital and more agility for pharmacists to authorize a personalized device to come into the OR.


In countries like France, where CMF cranioplasty personalized devices already account for 30% of all surgeries, the CE marking will bring considerable benefits in managing these devices within the hospital.
We expect that this certification will facilitate and encourage more physicians to treat patients with personalized solutions worldwide. In addition, the CE marking carries international recognition, which will also help scale the technology outside Europe.


To the best of our knowledge, we are the first company to have such a large, personalized portfolio that is CE-marked. Ultimately, we hope that more companies developing 3D-printed devices will follow this lead and acquire this kind of certification.
It takes a journey to scale personalization in healthcare
At Materialise, personalization is our core business. For the last three decades, we have worked closely with the medical community and regulatory institutions worldwide to collect data and validate the quality of 3D-printed devices as an effective and reliable production system, as trusted as any traditional technology.
The CE marking is an essential milestone we have crossed in the journey to scale personalization in healthcare, as it will help streamline access to these devices within hospitals.
As tech innovators and leaders, we know that this is one of the steps in the big challenge we have in front of us: making personalized solutions even more accessible, cost-effective, and sustainable for hospitals and users. This means we want to scale to a level of mass personalization.
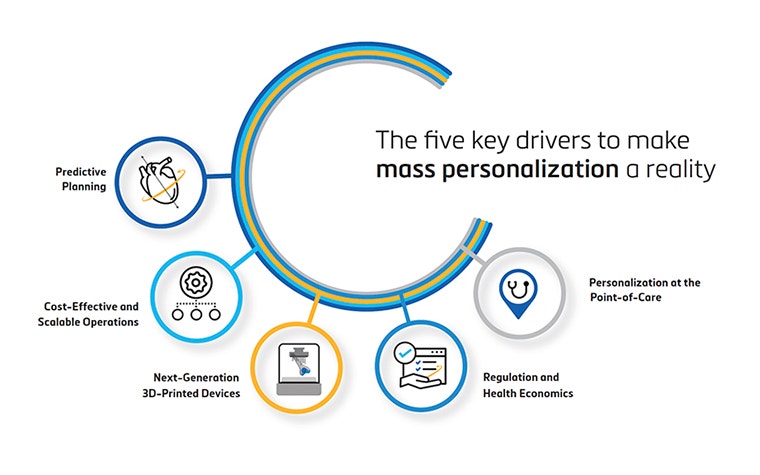
We know these ambitions are bold, and they need to be paired with action. We plan to tackle them through these five pillars:
- health economics and regulation
- next-generation 3D-printed devices
- cost-effective and scalable operations
- predictive planning
- personalization at the point of care
Our latest whitepaper details the progress on our commitments for each of those pillars. It also reveals some of the big lessons we have learned and provides more information about approaching these areas in the years to come.

Why do we need to talk about scaling personalized solutions?
In the last five years, we have seen significant growth in our solutions for 3D point-of-care facilities. This is not an innovation trend but a recognition of the cost benefits that a personalized approach brings in the long term. By now, most medical device companies have also incorporated customized solutions into their portfolio. Check out this interview with our partner Zimmer Biomet about the great potential of growing personalization in knee surgeries and its benefits to surgeons and patients.
Scaling personalized solutions has incredible potential to improve patient outcomes by enabling less invasive surgeries performed in a shorter time and done right the first time.
And as in the scalability process of any successful technology, there comes a crucial point when technology, availability, and an understanding of the market's needs all mature. This makes the technology not only feasible but unavoidable.
We believe we are now close to this inflection point with personalized surgical solutions.
If you are part of the medical community, we would love to hear your opinion on scaling to more personalized healthcare treatment in the near future.
L-102216-01
Share on:

Meet the author
Brigitte de Vet
You might also like
Never miss a story like this. Get curated content delivered straight to your inbox.
