EXPERT INSIGHT
3D-Printed Non-Invasive PEEP Masks Aim to Alleviate Ventilator Shortage
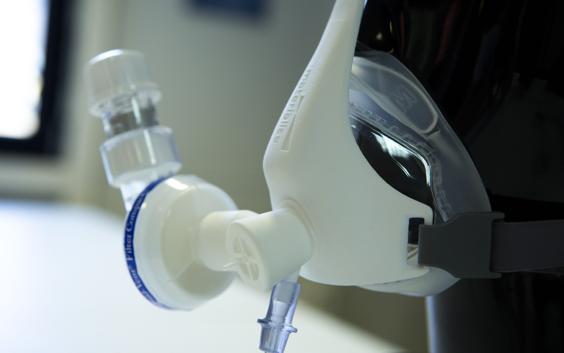
As coronavirus infections continue to rapidly spread, hospitals around the world are in dire need of the necessary medical equipment to treat patients. Mechanical ventilators are one of these essential items needed during this pandemic that are critically under-supplied.
As our mission is to create a better and healthier world, we set out to explore ways to help healthcare workers with the ventilator shortages. While many parties are increasing the production of these devices, we have looked at the problem from a different angle. We saw that clinicians are looking for other ways to deliver positive end expiratory pressure, also known as PEEP, to COVID-19 patients, and worked closely with them for an alternative.
We developed a solution to deliver oxygen and create high positive pressure without the use of a ventilator by designing a 3D-printed connector that holds together standard medical equipment, namely a non-invasive mask, a filter, and a PEEP valve. This 3D-printed solution for an adjustable PEEP ventilation is called the Materialise Passive NIP, with NIP standing for non-invasive PEEP, and is currently being fast-tracked through the regulatory process to make it available as soon as possible during this crisis. In parallel, we are supporting a clinical trial to define the best use of the new solution for COVID-19 patients.
Helping patients by delivering positive pressure
In most clinical settings today, physicians would switch from a pure oxygen mask to a ventilator. Prof. Wilfried De Backer, Pulmonologist and Director of respiratory imaging company FLUIDDA, who partnered with us on this development, notes, “The aim is to avoid invasive treatments. This new solution is giving positive pressure and oxygen, so it is not a big burden on the patient. So you can avoid more invasive treatments and save on ventilator capacity, which you can use on other patients.”
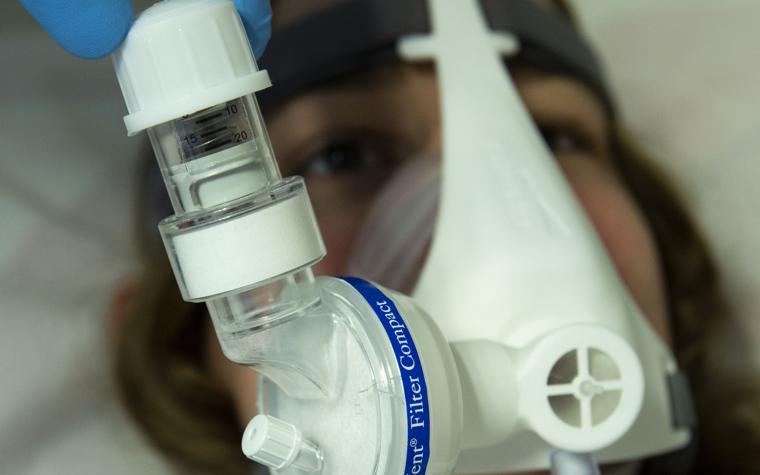

“The answer to the question 'when do we start with the mask?'” as Prof. De Backer continues, “is, I think, when the patient is getting worse and there is a higher need for oxygen. This is the ideal time to switch from a pure oxygen mask, where you just deliver oxygen, to a mask that also delivers this positive pressure in the airways, pushing back the fluids in the lung and allowing for a better uptake of oxygen. If we can give positive pressure, and we keep the situation stable, the patient stable, we can avoid more invasive steps.
“For the caregiver, because the mask fits well, meaning without a leak, and there is a filter added to the solution, there is not as much of a risk in spreading the virus in the environment.”
Fast and reliable
3D printing could have a significant role to play in the COVID-19 crisis, as some of its main strengths include speed and freedom of design. Plus, it allows for production to be done locally and in a safe, responsible way. To design this mask, we relied on our decades of 3D printing experience, along with our extensive medtech know-how, ISO 13485 certification, and expertise in developing medical devices in accordance with EU and US regulations.
Within a matter of days, our engineers were able to go from idea to proof of concept that was tested together with a pulmonologist. A few days later, the solution was ready for verification testing on healthy people.
In terms of the design itself, our engineers created the Materialise NIP Connector to be as efficient as possible. As our engineer Jan Van Espen explains, “It’s a very compact design. We have an oxygen supply for the patients, and through the one-way inhalation valve, we can take in extra ambient air. We have a filter to minimize aerial contamination with respect to the COVID virus, and we have an adjustable PEEP valve, which is the positive pressure for the patients.”
Another engineer working on the project, Ben Bekaert, notes it’s easy to set up: “Once assembled, you just have to connect the Passive NIP to the oxygen supply. It’s a simple system.”


Plus, because the solution was created using 3D design, it makes it possible to manufacture locally and, therefore, bring to hospitals quickly. This becomes even more crucial as travel and transport become more difficult. In this case, the product was designed in Belgium and can be printed at a Materialise-certified facility, or at a hospital that has the capability to do so in a reliable manner. Also, smaller production series sizes enabled by 3D printing allow designs to be customized to fit all types of NIV masks available at hospitals.
Clinical trial on COVID-19 patients
The Materialise NIP Connector and the designs are available. We have tested it on healthy people, and we are fast-tracking the regulatory registration in Europe and the US. At the same time, we are upscaling production to be able to deliver this broadly to the clinical community.
In parallel, we are supporting a clinical trial to determine the effect of the Passive NIP on COVID-19 patients. Our clinical partners are hoping to provide clinical guidance regarding the optimal use of the Materialise Passive NIP.
As Prof. De Backer notes, “Except for the new 3D-printed connector, the components of the mask exist, and the concept is already known. We’ve used it for years in non-invasive and invasive ventilation. In the trial, we will identify the effect on COVID-19 patients and at what stage of the disease to best use this NIP solution. We think that it is important to start very early, so just when the infection or the inflammation in the lower airways starts, and that at that time we switch from pure oxygen to this combined treatment with oxygen and with pressure.”
We realize that many hospitals are in dire need, so we will work with them to provide this as an emergency solution in compliance with local regulations. We are also open to partnering with others in order to accelerate access for patients around the world.
Collaboration is more important than ever as we face the global coronavirus pandemic and the associated shortage of ventilators and other medical equipment.
Share on:
You might also like
Never miss a story like this. Get curated content delivered straight to your inbox.
