PRESS RELEASE
New Software Supports Increased Demand for Personalized Medical Devices
December 2, 2020
Materialise SurgiCase Streamlines Collaboration Between Medical Device Companies and Clinicians
Leuven, Belgium, – December 2, 2020. Materialise NV (Nasdaq: MTLS),a global leader in 3D printing solutions, has released SurgiCase software, an online case management system (CMS) for personalized medical devices. The SurgiCase software, will give medical device companies and clinicians a single location to exchange data, communicate about procedural planning and check the status of their cases. This reduces the administrative overhead and ultimately helps these companies to help more patients with a personalized device at a lower cost.
The demand for personalized medical devices has grown significantly in recent years as physicians experience their benefits for predictable, accurate outcomes and improved patient care. This comes with new challenges for medical device companies. Instead of making one single device in a more efficient way, nowadays medical device companies need technology to create thousands of personalized devices in an automated workflow, while ensuring a good communication with the individual clinicians.
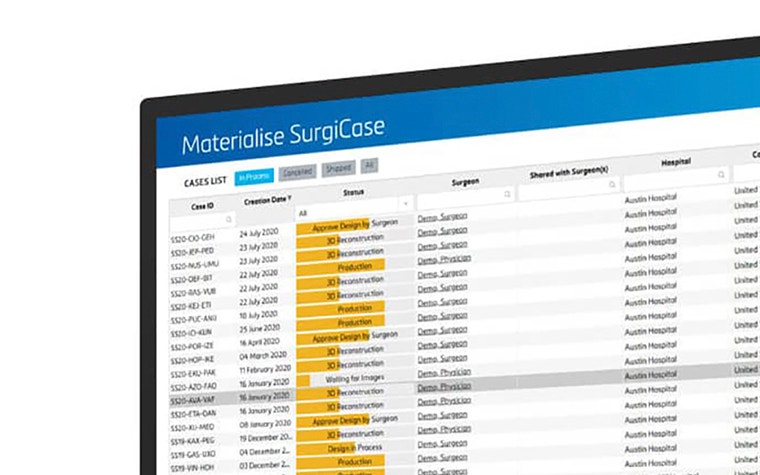

The Materialise SurgiCase software will help these companies optimize their logistical process by streamlining several steps in their planning and design process, from order intake and case tracking, to reporting and compliance checks. It will accelerate their case management by providing an overview of all ongoing cases, historical case data and file storage and approval management.
On the clinicians' side, SurgiCase improves the collaboration between physicians and clinical engineers by allowing physicians to inspect and approve planning and device design through an interactive viewer that combines 2D images and 3D models. Throughout the entire process the software allows physicians and staff to provide input regarding service, delivery date, case information, while tracking the progress of ongoing cases.
“Now personalized medical devices are moving from novel to mainstream, medical device companies are confronted with new operational challenges. From the need for an image upload platform, to a way to keep their customers informed on the status of their order,” said Sebastian De Boodt, Market Manager Medical Software, “For years, we’ve developed and perfected the SurgiCase software to increase the efficiency in our internal operations. By bringing it to the market as a software product, we want to empower other medical device companies to help even more patients with a personalized device.”
Materialise leveraged its 20 years of expertise in optimizing personalized device processes to power SurgiCase’s seamless case ordering and planning processes. The software is complementary to the Materialise Mimics Innovation Suite, the most advanced toolkit for engineering on anatomy, and can be used in an ISO13485 and GDPR compliant setting.
Materialise Medical has pioneered many applications of 3D printing and planning, enabling researchers, engineers, and clinicians to revolutionize innovative personalized treatment that improves lives. The company’s open and flexible platform of software and services which forms the foundation of certified medical 3D printing.
About Materialise
Materialise incorporates three decades of 3D printing experience into a range of software solutions and 3D printing services, which together form the backbone of the 3D printing industry. Materialise’s open and flexible solutions enable players in a wide variety of industries, including healthcare, automotive, aerospace, art and design, and consumer goods, to build innovative 3D printing applications that aim to make the world a better and healthier place. Headquartered in Belgium, with branches worldwide, Materialise combines the largest group of software developers in the industry with one of the largest 3D printing facilities in the world. For additional information, please visit: www.materialise.com
Materialise press contact
Share on: