Webinar
Making Pulmonary Interventions Truly Personalized: From Treatment Development to Patient Care
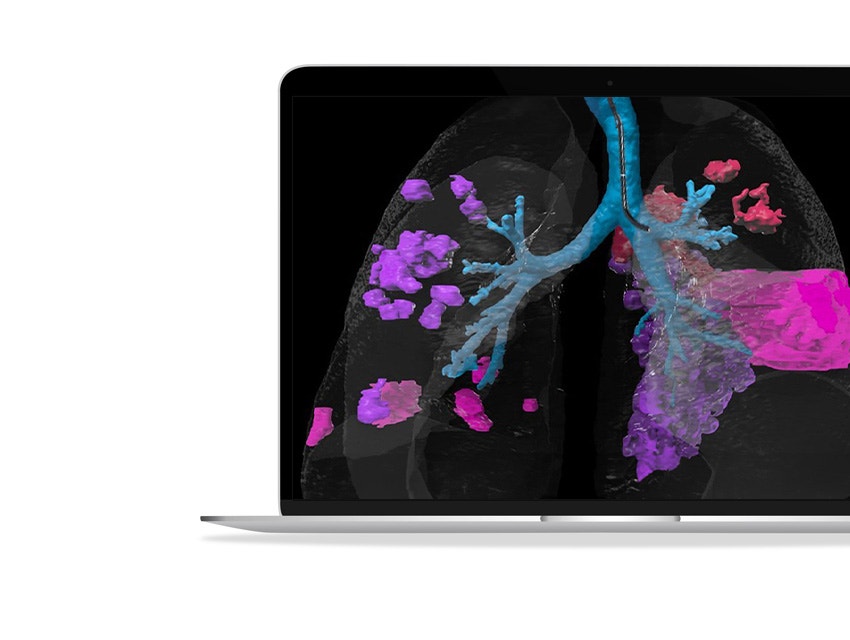
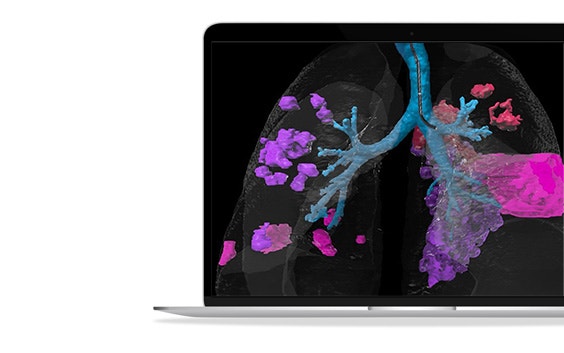
About this webinar
Are you a medical device company that is developing a novel respiratory treatment, or that has a treatment solution on the market?
In this webinar recording, you will discover how data-driven technologies like artificial intelligence can help optimize respiratory treatment protocols, resulting in better clinical trial outcomes, and how clinicians benefit from smart patient screening and 3D planning tools to better prepare for treatments and achieve more predictable results. This webinar is brought to you by Fluidda and Materialise, bringing together decades of expertise in advanced respiratory imaging and personalized 3D planning.
What you will learn
- How advanced CT analysis techniques uncover a deeper understanding of lung pathology and disease progression to define and optimize treatment parameters for your device
- How smart patient screening and 3D treatment planning can lead to more successful clinical trials, trial time reduction, and a guarantee of predictable outcomes
- How to cost-effectively scale your treatment planning when going from helping one patient to millions more
Speakers

Maarten Lanclus PhD

Antoon Dierckx
Share on:
Materialise medical device software may not be available in all markets because product availability is subject to the regulatory or medical practices in individual markets. In countries where no regulatory registration is obtained of Mimics or 3-matic Medical, a research version is available. Please contact your Materialise representative if you have questions about the availability of Materialise medical device software in your area.
L-102297
THINK | Idea to Patient Care
As an engineer in the medical field, your dream is to make a positive impact on patient care. To inspire you as you advance anatomical engineering even further, we have created a webinar series that sets the stage for our customers to share their powerful insights on how their cutting-edge work is contributing towards making the world a healthier place through innovation.
