Webinar
Bringing Personalized Medical Devices to the Masses: How Terumo Aortic Optimizes its Online Case Management
About this webinar
Personalized care is moving from novel to mainstream. However, there are still considerable hurdles, such as administrative overhead for physicians and operational inefficiencies for businesses when it comes to adopting personalized devices.
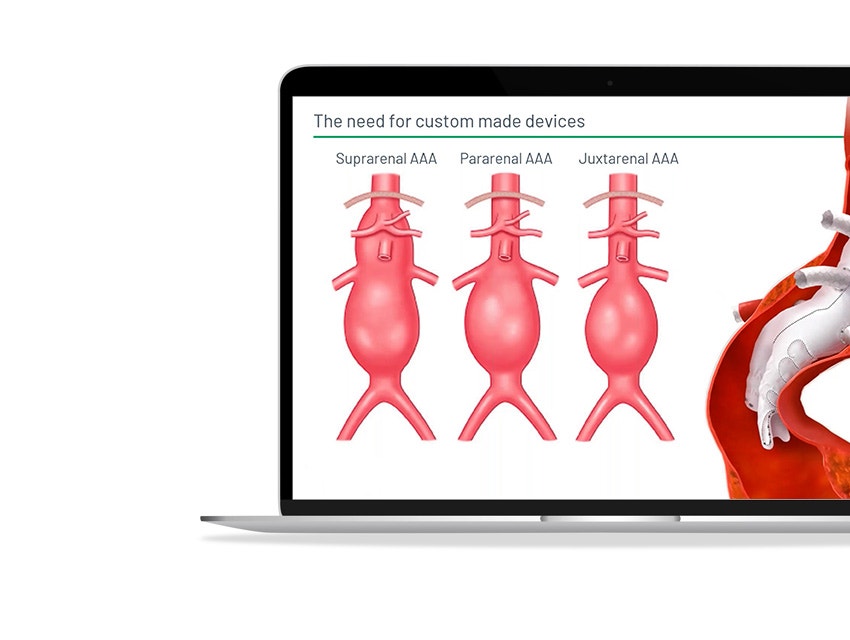
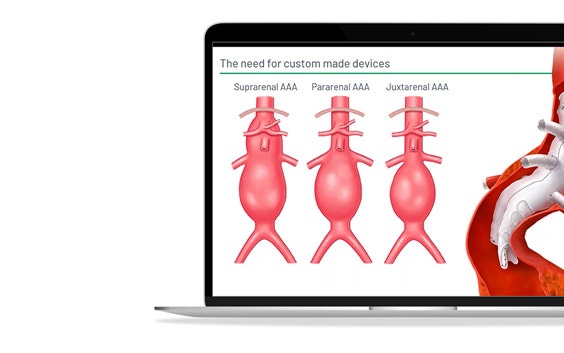
Terumo Aortic, a medical device company specialized in developing aortic and peripheral vascular disease solutions, also faced these challenges when scaling up its custom stent-graft business. In this webinar, they explain how they integrated Materialise SurgiCase, an online case management platform, into their clinical operations to enhance physician experience and boost operational excellence.
What you will learn
- The challenges the Terumo Aortic team faced when adapting to the increasing demand for their personalized devices
- How Materialise SurgiCase supported them in scaling up their personalized device business and streamlining their operations effectively
- How they improved their customer experience and collaboration with physicians through an easy-to-use communication platform
Speakers

Bastien Pirson

Jamie McCarte
Share on:
Materialise medical device software may not be available in all markets because product availability is subject to the regulatory or medical practices in individual markets. In countries where no regulatory registration is obtained of Mimics or 3-matic Medical, a research version is available. Please contact your Materialise representative if you have questions about the availability of Materialise medical device software in your area.
L-102232
THINK | Idea to Patient Care
As an engineer in the medical field, your dream is to make a positive impact on patient care. To inspire you as you advance anatomical engineering even further, we have created a webinar series that sets the stage for our customers to share their powerful insights on how their cutting-edge work is contributing towards making the world a healthier place through innovation.
