CUSTOMER STORY
Micro-CT Imaging Reconstructs the Tree of Life: Understanding Placental Blood Flow
We’re pleased to introduce Nikhilesh Bappoo, our first prize winner of the Mimics Innovation Award 2017 APAC region. The Mimics Innovation Awards have existed since 2005 and have celebrated many researchers who have demonstrated innovation and excellence in the field of medical technology. The reward offers international recognition as well as a monetary prize to help fund a healthy continuation of the research. Participants using Materialise Mimics in diverse environments, such as hospitals, medical device companies, and academia are all welcome to participate.


Nikhilesh and his research
It was under the supervision and guidance of Dr. Barry Doyle, Head of Vascular Engineering Laboratory at the Harry Perkins Institute of Medical Research, and Dr. Caitlin Wyrwoll from the Reproductive Biology Laboratory at the School of Human Sciences, University of Western Australia (UWA), that Nikhilesh started working on his honors project in 2016.
Nikhilesh’s experience includes tutoring at the UWA for a biomedical engineering course, helping students on how to use Mimics and 3-matic as part of their patient-specific design project. An invitation followed to present his winning paper at the International Federation of Placental Associations conference in Manchester, UK. Winning the award also brought him into contact with the Materialise Malaysia Office, where he was accepted to do an internship in the DLP Competence Centre.
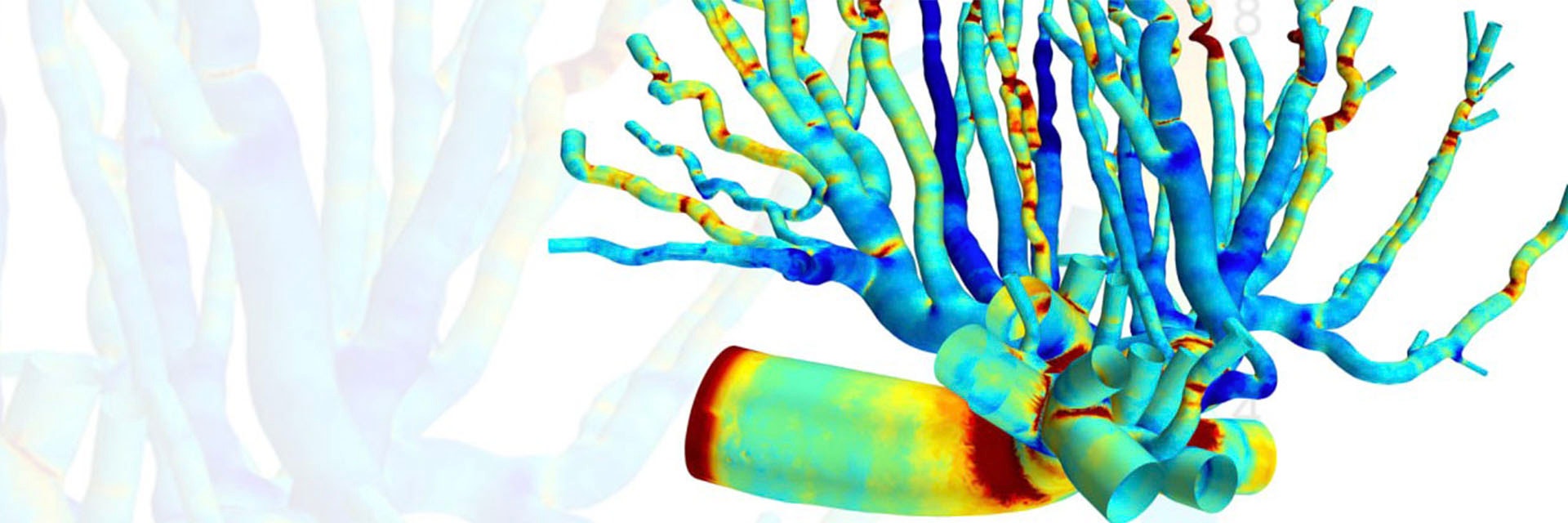
Nikhilesh developed a bioengineering framework to better understand how evolving blood flow affects the biomechanics of placental vasculature during pregnancy. In his groundbreaking research paper, Nikhilesh explores placental physiology by creating a 3D computational fluid dynamics (CFD) model of a rat’s placenta. With this complex vascular modeling, he further investigates biomechanical forces in the arterial network. He uses mechanical parameters such as wall shear stress (WSS), the pressure created against the arterial walls when blood flow velocity changes, to explain how blood vessels respond to blood flow and therefore develop into the fundamental complex tree structure required to support a healthy fetus.


Why study placental vascular biomechanics?
As pregnancy progresses, blood flow changes due to the increasing needs of the growing fetus. Chances of the placenta not responding to these changes in blood flow can be higher in some women and lead to complications during their pregnancy. Understanding this type of change in a numerical way is quite unique and could lead to very interesting findings and provide better diagnostic tools and better treatment plans.
"Currently, there is a lack of research concerning placental physiology," writes Nikhilesh in his paper. There isn’t enough computational hemodynamic data to help us understand this ephemeral organ. This mysterious, complex geometry is also what makes the highly detailed results of this study obtained by micro-computed tomography (µ-CT) so interesting. Nikhilesh is thinking of ways to translate more similar results into clinical use for the future.
Using 3D modelling and CFD
In his research, Nikhilesh was trying to understand how fluid biomechanics influences the way the geometry of the blood vessel network is formed, as well as where and to what extent WSS occurs.
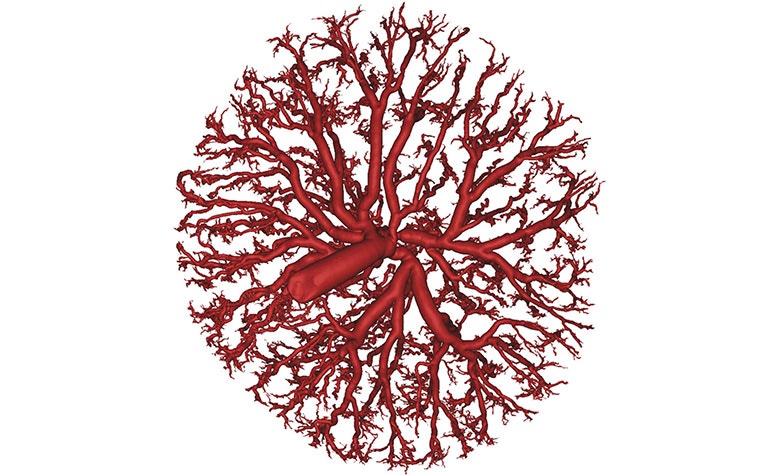
A female rat in late pregnancy was used for the research, as there is much resemblance between human and rat placentas. The feto-placental arteries were perfused with Microfil and scanned via micro-CT imaging. Nikhilesh used Materialise Mimics to convert the micro-CT images into 3D models of the arterial structure, using segmentation tools like thresholding and region-growing. He used Materialise 3-matic to manually remove small defects in the model. He then used a first order Laplacian smoothing to remove noise on the exterior and interior surfaces.

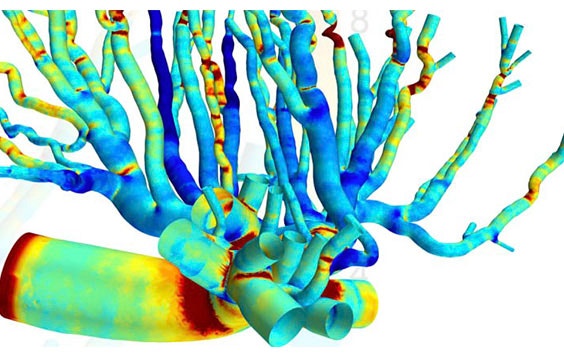
“The reconstructed geometry was an incredibly beautiful, complex, and detailed network of blood vessels. Mimics provides a new dimension for researchers, clinicians, and bioengineers to understand the anatomy of the most complex organs, such as the placenta.”
— Nikhilesh
Nikhilesh created a sub-volume of the network in order to reduce the size of the model for computational fluid dynamics, and used the centerline tools in Mimics to obtain a path for the blood flow for each vessel. He then exported the models from Mimics and used STAR-CCM+ from Siemens to create computational meshes.
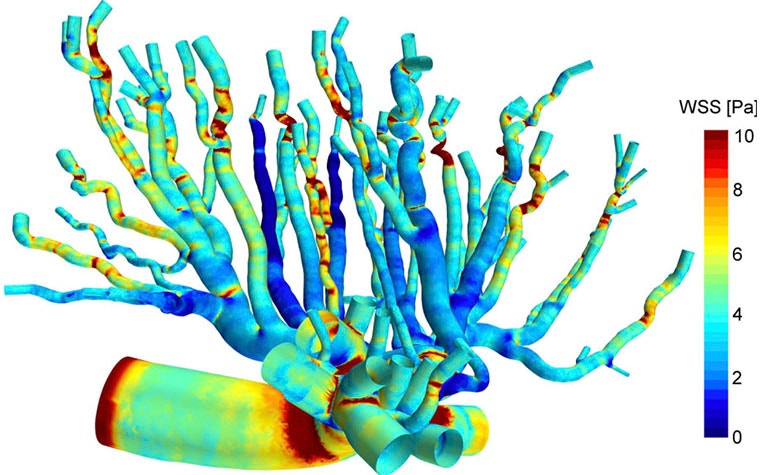

Findings of the research
The CFD simulation he ran shows that where bifurcation occurs, frictional forces are the strongest. These biomechanical blood forces dictate how the vascular tree structure will develop to meet the evolving needs of the fetus during late pregnancy, says Nikhilesh, adding that these patterns can now help us further understand the physiology of the placenta.
This work represents the innovative beginning of understanding placental blood flow and how this could later be translated into clinical applications. Applying 3D modelling to the placental blood flow of a human placenta could help better predict its vascular structure expansion and even prevent pathological disorders in pregnancy.
The placenta’s ephemeral state has long been the subject of folklore in many cultures. Although this innovative research is still in its infancy, the knowledge it already provides in the field of placental hemodynamics sets the foundation for a comprehensive understanding of the placental vascular network. While we can still be in awe at the placenta’s vital role during pregnancy, eat it as part of a ritual, plant it under a tree, or simply discard it after birth, science and mathematics can start clarifying its mysteries.
Nikhilesh will start his PhD this year. His Mimics Innovation Award grant will hopefully contribute to furthering research on placental hemodynamics and help translate the results into clinical applications.
Share on:
You might also like
Never miss a story like this. Get curated content delivered straight to your inbox.