CASE STUDY
Creating the Perfect Fit for a Pulsatile Total Artificial Heart
The accurate fit of a total artificial heart (TAH) is an important challenge since every patient’s anatomy is unique. German device engineers were looking for a way to optimize their designs to treat the maximum number of patients.

Industry
Healthcare
Key solutions
Materialise Mimics
The impact
Larger study population
Consistent measurements
Virtual design iterations
The challenge
A total artificial heart (TAH) replaces the human heart both functionally and anatomically. Since the device is implanted permanently into a patient, the accurate fit of this device is an important challenge. However, due to the technical complexity and cost of such devices, only a limited number of designs are economically feasible.
This challenges device engineers to find a way to optimize their designs and ensure compatibility with the maximum number of individual patient anatomies.
For the design of the Aachen TAH ReinHeart, A.J. Fritschi and his colleagues at the Institute of Applied Medical Engineering at RWTH-Aachen University in Germany sought out Materialise Mimics software to create anatomical models from real patient data in order to determine fit and reveal areas for improvement.
The major challenge in this type of study is handling the large volume of medical imaging data. All patient data must be processed in precisely the same way to ensure comparability of the findings and, thus, the credibility of the study. These requirements demand a highly accurate workflow and a significant time investment due to a large number of patients.
The solution
Virtual implantations in 27 patients using Mimics
Fritschi et al. selected medical images from 27 patients as the input for their design validation. They segmented these datasets to create detailed virtual models in Mimics. With the help of the software, all 3D models were aligned using a common coordinate system to derive meaningful measurements from the virtual analysis (Figure 1).
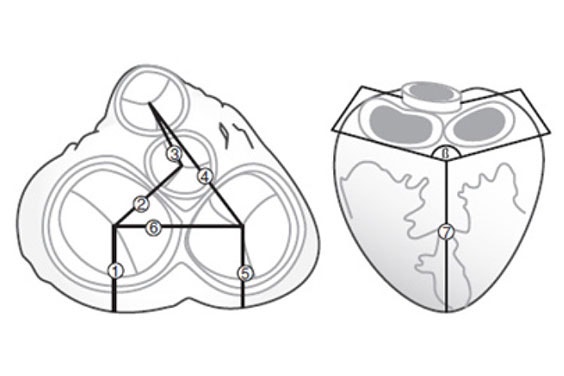
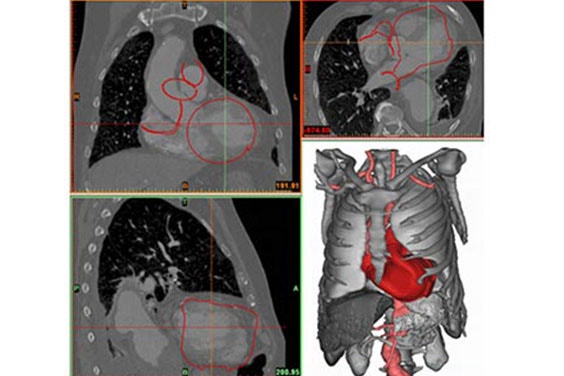
Figure 1 and Figure 2
Fritschi et al. ensured the reproducibility of the model creation by using tools that can be applied globally to the images, such as thresholding, region growing, and morphological operations (Figure 2). The authors were interested in determining the spatial constraints of the pericardial cavity and the valve position and orientation. They used Mimics to measure oblique parameters according to anatomical landmarks (Figure 1). All measurements made were comparable between patients.
In the next step, Fritschi et al. performed a virtual fitting of the current artificial heart design to each of the individual patients’ 3D models (Figure 3). This virtual fitting is an accurate and cost-effective way to determine the precise device fit and reveal areas for improvement.
Figure 4 shows an overlay of the CAD model of the artificial heart on a patient’s heart. The alignment could also be verified using the original image data (Figure 3), which allowed the authors to iteratively improve the device design.
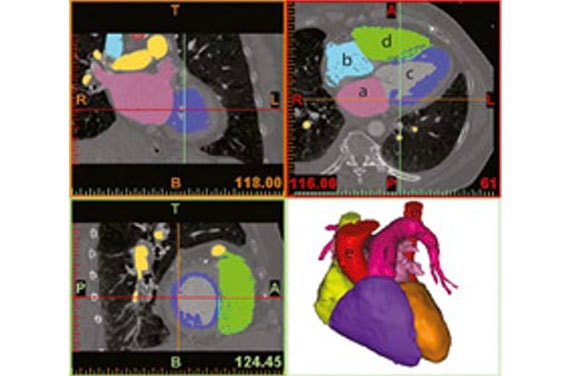
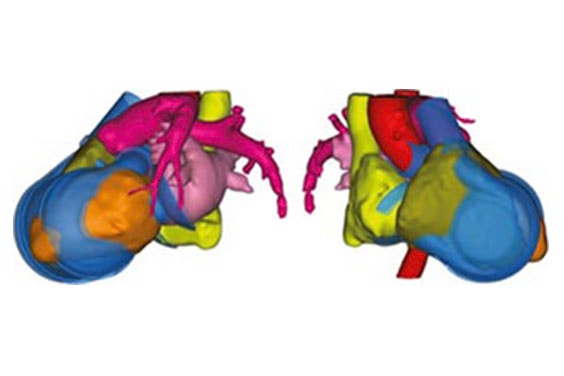
Figure 3 and Figure 4
Finally, the researchers verified their virtual fitting process through cadaver studies, and a 3D capturing system acquired the position of the implanted artificial heart (Figure 5). This information was transferred to Mimics, where a further design analysis was performed.
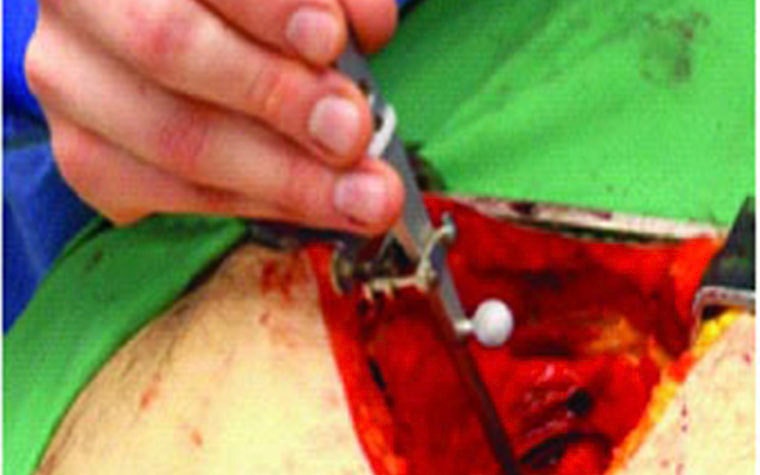

The result
Validation that virtual fitting plays a valuable role in device design
Fritschi et al. concluded that virtual fitting is an effective tool for device design and an economical solution for including a larger number of patients in the study. The authors caution, however, that conventional cadaver studies will still play an important role, as many key elements of surgery, such as haptic feedback, are not yet available in virtual studies.
This study, which was published in BioMedical Engineering OnLine, earned Fritschi et al. a Mimics Innovation Award, which celebrates cutting-edge work by early-stage researchers using Materialise software to advance personalization in healthcare.
Share on:
