Webinar
Automating Key Components of Finite Element Research
About this webinar
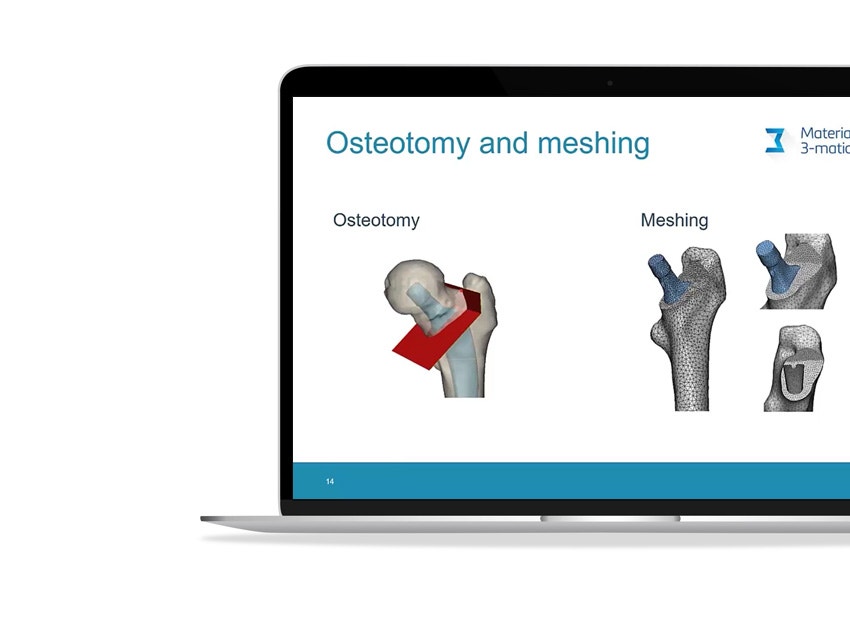
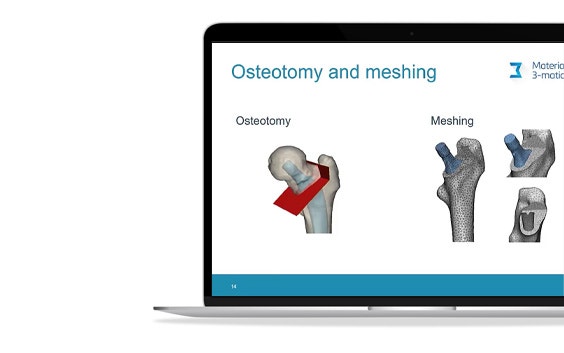
In total hip replacement surgery, most traditional designs of femur components have a long stem. Short hip stems have only recently been introduced, and these are presumed to reduce proximal stress shielding compared to traditional, long stems.
However, due to their smaller contact area with the bone, high peak stresses and areas of stress shielding could appear in the proximal femur, especially in the presence of atypical bone geometries. Researchers at the University of Leuven and the University Hospital of Brussels wanted to better understand this aspect by virtually implanting a commercially available calcar-guided short stem (Optimys from Mathys AG, Bettlach, Germany) in a series of bones with deviating proximal geometry, and by performing finite element analyses with the help of automation.
What you will learn
- How researchers automated time-consuming, labor-intensive work to assess hip implant performance
Speakers

Ir. Amelie Sas
Share on:
Materialise medical device software may not be available in all markets because product availability is subject to the regulatory or medical practices in individual markets. In countries where no regulatory registration is obtained of Mimics or 3-matic Medical, a research version is available. Please contact your Materialise representative if you have questions about the availability of Materialise medical device software in your area.
L-100770-01
THINK | Idea to Patient Care
As an engineer in the medical field, your dream is to make a positive impact on patient care. To inspire you as you advance anatomical engineering even further, we have created a webinar series that sets the stage for our customers to share their powerful insights on how their cutting-edge work is contributing towards making the world a healthier place through innovation.
