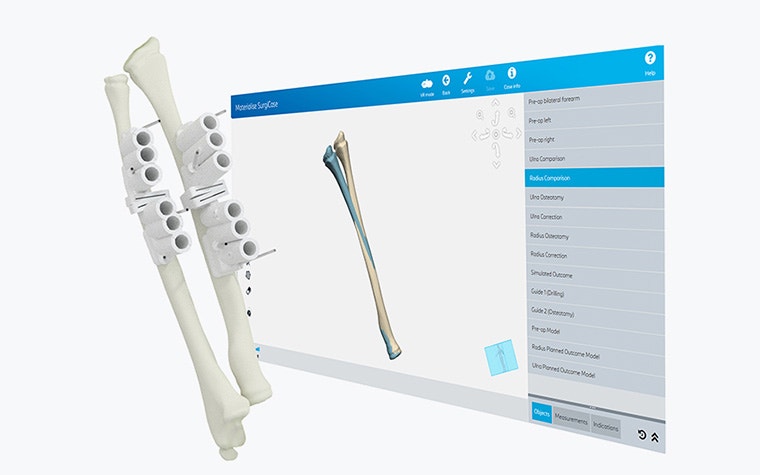
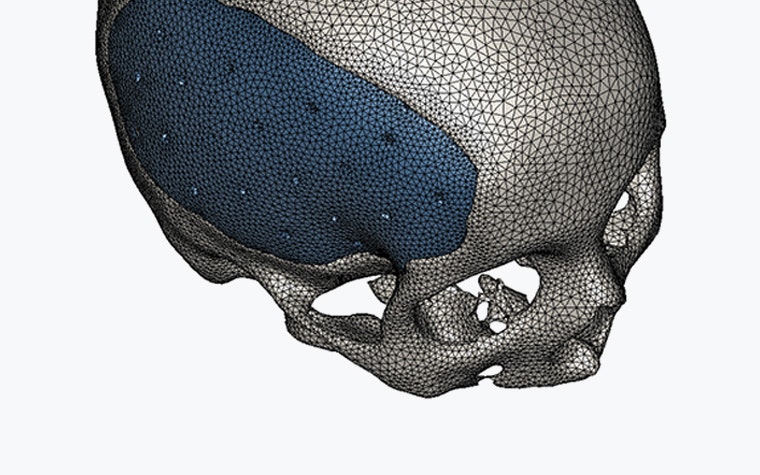
Our software solutions are registered as medical devices in many countries around the world. This confirms their safety and effectiveness, as well as the quality standards they meet.
In some regions, customers can refer to our Master File kept by the local regulatory body. This file holds most of our DHF documentation so the regulatory body can compare your workflow to ours. For example, they can refer to our verification and validation process for segmentation accuracy.
A research version is available in countries where Mimics and/or 3-matic Medical have not obtained regulatory registration. Please contact your Materialise representative if you have questions about the availability of Materialise medical device software in your area.